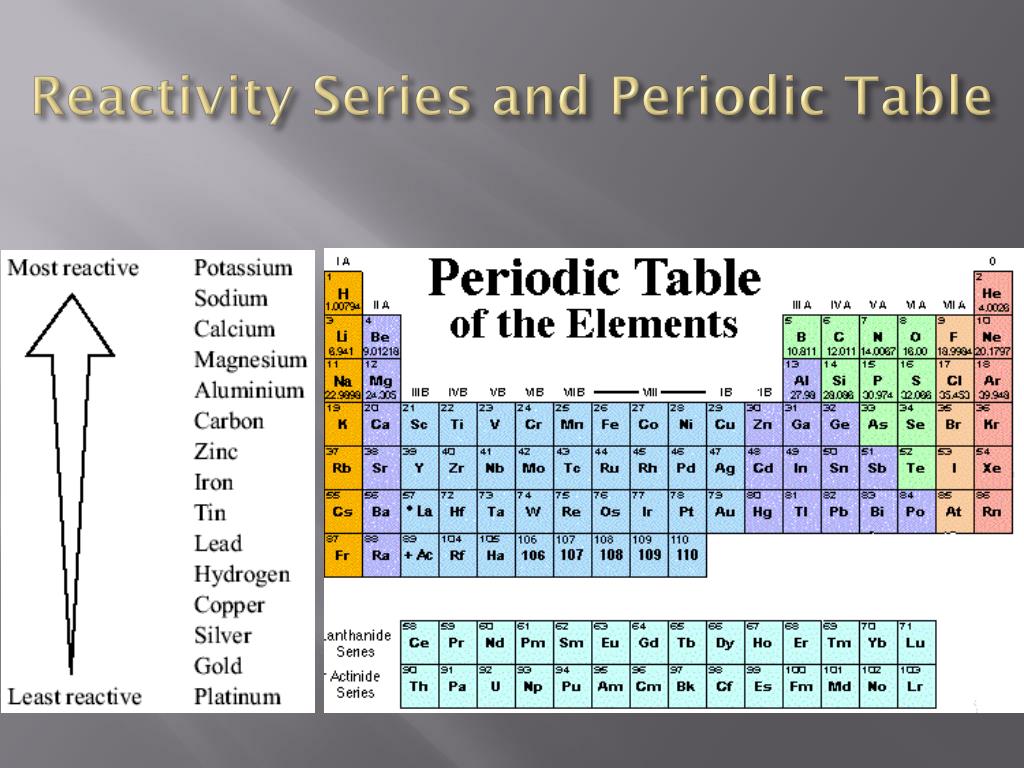 
For example, all the elements in group 1 are reactive metals, and all the elements in group 0 are unreactive non-metals. You can effortlessly find every single detail about the elements from this single Interactive Periodic table. The modern periodic table is based closely on the ideas he used. The 1 st period has only two elements: hydrogen in group 1 and helium in group 18, with a gap from group 2 to group 17. This above image clearly shows you where are Alkali metals located on the Periodic table. Alkali metals include: Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs) and Francium (Fr). The periods are numbered: 1 at the top to 7 at the bottom. Alkali metals are located in group 1 on the left side of the Periodic table. Simple Chemical Reactions Unit Test 3 of 193 of 19 Items Question Use the periodic table to answer the question. The periodic table has seven horizontal rows called periods. The alkali metals are so called because reaction with water forms alkalies (i.e., strong bases capable of neutralizing acids). Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Top answer: The two groups of elements in the periodic table that are the most reactive are the alkali metals in Read more. alkali metal, any of the six chemical elements that make up Group 1 (Ia) of the periodic tablenamely, lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. Note: Astatine (atomic number 85) shows characteristics of nonmetals (halogens) as well as metalloids. All these nonmetals are located on the upper right corner of the Periodic table ( Hydrogen is located on the left top corner) In the above image, the nonmetals are represented in yellow color. These metals are known as alkali metals and include elements such as lithium, sodium, and potassium. There are 18 nonmetals on the Periodic table. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). Reactive nonmetals are a group of chemical elements that are located in the upper right-hand corner of the periodic table.This group includes 11 elements, namely hydrogen (H), carbon (C), nitrogen (N), oxygen (O), fluorine (F), phosphorus (P), sulfur (S), chlorine (Cl), selenium (Se), bromine (Br), and iodine (I). The most reactive metals are located in the lower left of the periodic table. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Other chemical properties include: Electropositive Character: Metals tend to have low ionization energies, and typically lose electrons (i.e. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
